This symphony of cracks and clicks makes you feel as though
you can reach any key and can work faster and more dexterously than
you could’ve before. Will you sound better now that you have cracked your
knuckles? Nope – you don’t play piano; you’re here to move it to the next room.
Question of the Day –
What makes the sound when you crack your knuckles and does it help or hurt you?
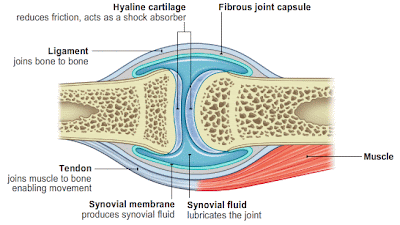
A knuckle is the
joint where your carpal meets your metacarpal. A joint is any hinged meeting of
two bones, made up of a sac (bursa) that keeps everything together and a small
space between the bones (synovial space). Fluid in the space between the bones
(synovial fluid) keeps the friction low as the bones flex in the joint.
Cracking joints can be done many at once or one at a time.
Big joints can be cracked just as smaller joints. Chiropractic practices make a living
out of cracking joints. There is an immediate feedback in hearing the joint
crack; some treatment must have been rendered.
No matter how or why the joints are cracked, the production
of the noise is the same in each case. Manipulation of the joint stretches the
joint, separating the two bones, and creating a larger space than normal.
In physics, pressure is related to volume, being
inversely related. The synovial joint has a certain amount of fluid, enough to
fill the normal space. By increasing the joint space volume, the fluid will be
filling a larger space. The same amount of fluid in a larger space means that
the fluid will be under lower pressure.
This decreased pressure in your closed joint will affect the
dissolved gases in the synovial fluid. All fluids of the body, like blood or
other extracellular fluid, even saliva, contains many molecules, including dissolved
gases; CO2, N2, O2. Gases in solution are
under pressure just like in their gaseous state. When the pressure decreases,
the amount of gas that can stay in solution decreases (it becomes less soluble). When gas becomes insoluble,
it comes out of solution and begins to form small bubbles (ebulism, a gas embolism). It is the formation of the bubble(s) that you hear.
Cavitation (formation
of a cavity) in small joints wouldn't seem to be a high-energy event, and it
isn’t, but it is enough to make the noise you hear. There is still some
question as to how such a large noise can be made this way, but x-rays can show
the presence of gas bubbles in the popped joint immediately after cracking.
After cracking your joint, the joint remains a little larger
for a period of time, and slowly returns to its normal volume. During the time
period that the joint is returning to the normal volume, pressure slowly
increases. More pressure means more gas solubility, and the bubbles disappear. It
takes a while, so you can’t crack that knuckle again for 15-20 minutes.
Is it bad for you to crack your joints? Does it do damage to
your joints, either immediately or over time? There hasn’t been a lot of
research done in this area, but what has been done shows that knuckle cracking
does not lead to osteoarthritis.
A 2011 study is the most comprehensive done to date. These
researchers looked at cracking and the frequency of cracking as well. No amount
of cracking seemed to promoted arthritis development. A 1990 study stated that
knuckle crackers were more likely to also have small amounts of hand
inflammation and lower grip strength. However, this study could not conclude
that knuckle cracking caused the inflammation or loss of grip strength.
Oxygen’s counterpart, carbon dioxide, is also present in the
blood, on its way back to the lungs to be exhaled. Most of the CO2
is locked up as part of carbonic acid (H2CO3) or its
conjugate base, bicarbonate (HCO3), and this helps to maintain the
pH balance of your blood. Yet there is a little free CO2 dissolved
in your blood as well.
The major dissolved gas in your blood is N2,
nitrogen gas. Remember that air 80% nitrogen. This also happens to be the most
soluble gas in your blood, so more of your body’s allotment of nitrogen is
carried this way; less need for carrier molecules like hemoglobin or
bicarbonate.
This is all well and good until the pressure on your body
changes, like when you go scuba diving. Water weighs much more than air, so for
every 10 m (33 ft) you descend in the water, the pressure on your body doubles.
With more pressure, more gas will be soluble in the blood. This is the opposite
reaction from when you stretch your joints while popping your knuckles.
The increased gas volume dissolved in your blood is no
problem as long as you allow it to dissipate slowly. But if you have been at
depth for some time, and then you ascend too quickly, your body doesn’t have
time to adjust to the change in pressure.
The return to normal pressure means less gas will be soluble
in your blood. Where is all the gas you added to your blood by diving deep
going to go? It’s going to come out of solution and form bubbles. This is
decompression sickness, sometimes called the bends.
In the meantime, you are doubled over in pain (“the bends”).
Pain is one thing, but if a bubble in your blood happens to get stuck
somewhere, that’s called a gas embolus.
Nothing downstream of the bubble is going to be getting oxygenated blood, and
that means it will die. If it is
in heart vessel, that causes a heart attack, if it’s in your lungs capillary
beds, that’s a pulmonary embolism, if it is in your brain, that’s a stroke. Any
of these can kill you.
The best way to treat the bends is to prevent them. You must ascend
in stages, allowing time to adjust to the lower pressure at each depth. Your body
will take the excess gas out of the blood if given time. When you learn to
dive, much time is spent on the math involved in preventing decompression
sickness; if you have been at such a depth for so long, you will need to ascend
in X number of stages, with Y minutes at each stage depth.
If you don’t follow this, you’re in for a great deal of pain
and a trip to a decompression chamber. In the chamber, they will pump in extra
air to increase the pressure on your body, just like being at depth again. This
will put the gas back into solution. Then they will release the pressure, a
little at a time, allowing your body to take the excess gas out of your blood;
the equivalent of a staged ascension for depth.
Even pressurized airliners have lower than normal air
pressure (your ears pop), so no one is advised to fly after diving at depth for
at least 24 hours. Dives that don’t require a staged ascent should still be completed
at least 12 hours before flying. If you go diving in the morning, get on a
flight immediately afterward, and then start popping your knuckles – could your
hand explode?
Next week, another question in biology - can bacteria change the earth - the whole earth?
Blatteau, J., Brubakk, A., Gempp, E., Castagna, O., Risso, J., & Vallée, N. (2013). Sidenafil Pre-Treatment Promotes Decompression Sickness in Rats PLoS ONE, 8 (4) DOI: 10.1371/journal.pone.0060639
deWeber, K., Olszewski, M., & Ortolano, R. (2011). Knuckle Cracking and Hand Osteoarthritis The Journal of the American Board of Family Medicine, 24 (2), 169-174 DOI: 10.3122/jabfm.2011.02.100156






Pretty good post. I just stumbled upon your blog and wanted to say that I have really enjoyed reading your blog posts. Any way I'll be subscribing to your feed and I hope you post again soon. Big thanks for the useful info. Energy Dais presents the most comprehensive list of Drilling Companies in India, Middle East, USA & Canada. Choose from a list of 100+ handpicked Offshore Drilling Service providers to get custom quotations. list of drilling companies
ReplyDeleteAs Many Exceptions As Rules: Gas, Knuckles, And The Little Blue Pill >>>>> Download Now
Delete>>>>> Download Full
As Many Exceptions As Rules: Gas, Knuckles, And The Little Blue Pill >>>>> Download LINK
>>>>> Download Now
As Many Exceptions As Rules: Gas, Knuckles, And The Little Blue Pill >>>>> Download Full
>>>>> Download LINK Cz
This comment has been removed by the author.
ReplyDeleteNice work, oh wise one. Yes!
ReplyDeleteInformative and serious, yet light and entertaining.
- JN
As Many Exceptions As Rules: Gas, Knuckles, And The Little Blue Pill >>>>> Download Now
ReplyDelete>>>>> Download Full
As Many Exceptions As Rules: Gas, Knuckles, And The Little Blue Pill >>>>> Download LINK
>>>>> Download Now
As Many Exceptions As Rules: Gas, Knuckles, And The Little Blue Pill >>>>> Download Full
>>>>> Download LINK St